Apr 9, 2019 — For the reaction nh4cl nh3 hcl at 25*c enthalpy change ∆h = +177 g mol-1 and entropy change ∆s=+285 jk-1mol-1 calculate free energy ...
Sep 21, 2010 — Here is the problem we are given: Calculate the standard enthalpy of formation for the reaction HCL (g) + NH3 (g) --- NH4CL (s), given .... NH4Cl as the sole product: HCl(g) + NH3(g) → NH4Cl(s). 1. Predict the ... Do not use published heats of formation, free energies of formation, or enthalpies.. Transcribed image text: Determine the enthalpy of the following reaction: NH4Cl (s) + NH3 (g) + HCl (g) Using the following information (fill in the blanks from the .... ∆H4 is obtained via mathematical manipulation of four equations and the enthalpy changes corresponding to the four related reactions: (5) NH3(aq) + HCl(aq) → .... NH4Cl (s) → NH3 (g) + HCl (g) · Stoichiometry · Enthalpy of Reaction · Entropy Change · Free Energy of Reaction (at 298.15 K) · Equilibrium Constant, K (at 298.15 K).
enthalpy
enthalpy, enthalpy formula, enthalpy equation, enthalpy definition, enthalpy symbol, enthalpy chemistry, enthalpy change, enthalpy calculator, enthalpy of reaction, enthalpy unit, enthalpy vs entropy, enthalpy of formation, enthalpy meaning, enthalpy of fusion, enthalpy of vaporization dibac for sketchup crack torrent
NH3(g)+HCl(g)⟶NH4Cl(s). Given the standard enthalpies of formation (below), calculate the enthalpy change for the reaction in kilojoules.. Click here to get an answer to your question ✍️ For the reaction NH4Cl(S) → NH3(g) + HCl(g) at 25^0 C, enthalpy change = + 177 kJ mol^-1 and entropy ... wiggle-text-art

enthalpy change
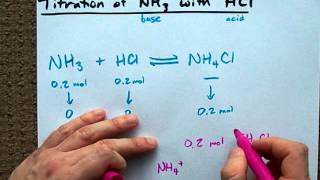
Sep 15, 2015 -- Questions. Chemistry. Calculate the enthalpy change (ΔH) for: HCl(aq) + NH3(aq) → NH4Cl(aq) The reaction of 50.0 mL of 1.00 M HCl with .... [SOLVED] Enthalpy of a Reaction Homework Statement Homework Equations So we are studying the reaction NH4Cl (s) ---> NH3 (g) + HCl (g) ... Download file photoshop-cc-2019-one-fundamentals.part1.rar (1,58 Gb) In free mode | Turbobit.net
enthalpy change of combustion
For the reaction NH3(g) + HCl(g) → NH4Cl(s): First, determine the enthalpy (ΔH°) and entropy (ΔS°) of the reaction. Enthalpy .... Oct 30, 2020 -- Using standard heats of formation, calculate the standard enthalpy change for the following reaction. NH4Cl(aq)NH3(g) + HCl(aq). 1. Expert's .... For any chemical reaction, the change in enthalpy in going from reactants to products is ... ie, NH3(g) and HCl(g) on the left, NH4Cl(s) on the right hand side.. Problem: For the reaction NH4Cl (s) → NH3 (g) + HCl (g) ΔH ° = +176 kJ and ΔG ° = +91.2 kJ at 298 K. What is the value of ΔG at 1000 K? a) -109 kJ b) -64 kJ c) .... What is the standard enthalpy of formation of NH4Cl(s) in the equation HCl(g) + NH3(g) = NH4Cl(s). A scientist measures the standard enthalpy change for the .... Reaction 1: HCl(aq) + NaOH(aq) --> NaCl(aq) + H2O(l). 50.0 mL 2.0 M ... Reaction 3: NH3 +HCl --> NH4Cl ... Calculate the Enthalpy of Reaction, delta Hrxn. 1.. Chemistry. The reaction of hydrochloric acid (HCl) with ammonia (NH3) is described by the equation: HCl + NH3 → NH4Cl A student is titrating 50 mL of 0.32 M .... Enthalpy change for the decomposition of ammonium chloride. 1. Write out the reaction NH4Cl(s) -> NH3 (g) + HCl(g) as a series of steps which include the ... 3e88dbd8be Scarbee Mm Bass Amped Crack Activate

